Latest News
Stay up to date with the latest research, product updates, events, and announcements from Ludger.
Oxford, 6th March 2026
PhD Position Available – GLYCOCALYX Doctoral Network (Marie Skłodowska-Curie)
Ludger Ltd is pleased to announce a fully funded PhD position within the Horizon Europe Marie Skłodowska-Curie Doctoral Network GLYCOCALYX.
The successful candidate will work on “Analysis of glycocalyx glycans using novel LC-FD-MS chromatography techniques”, developing advanced analytical methods to characterise the complex glycans that form the mammalian glycocalyx. The project focuses on improving high-resolution glycan analysis using innovative LC-FD-MS workflows, including approaches to distinguish biologically important sialic acid linkages.
The position will be based at Ludger Ltd (Abingdon, UK) with PhD enrolment at the University of Porto, and includes collaboration with partners across the GLYCOCALYX network, including research stays at i3S in Portugal.
The GLYCOCALYX Doctoral Network brings together 15 leading European academic and industrial partners to train PhD researchers in multidisciplinary approaches spanning chemistry, physics and biology to better understand the structure and barrier functions of the mammalian glycocalyx.
Application deadline: 3 April 2026 (17:00 UK time)
Location: Ludger Ltd, Culham Campus, Abingdon, UK
Applications: daniel.spencer@ludger.com.
For further details about the project and the GLYCOCALYX doctoral network, please visit: https://www.glycocalyx.org/phd-projects
Oxford, March 2026
Glycomic Markers Show Promise for Predicting COVID‑19 Outcomes
We are pleased to announce the publication of “Total plasma N‑glycomic patterns of COVID‑19 disease” (Elgood-Hunt et al., Glycoconjugate Journal), funded by Innovate UK (69371, 2021) and delivered in collaboration with Warwick Medical School, RCSI, and University College Dublin.
This landmark study analysed over 500 plasma samples from COVID‑19 patients across discovery, replication, and validation cohorts to map disease‑related glycomic changes at the population scale. The research identified distinct N‑glycan signatures associated with disease severity, ICU admission, and mortality.
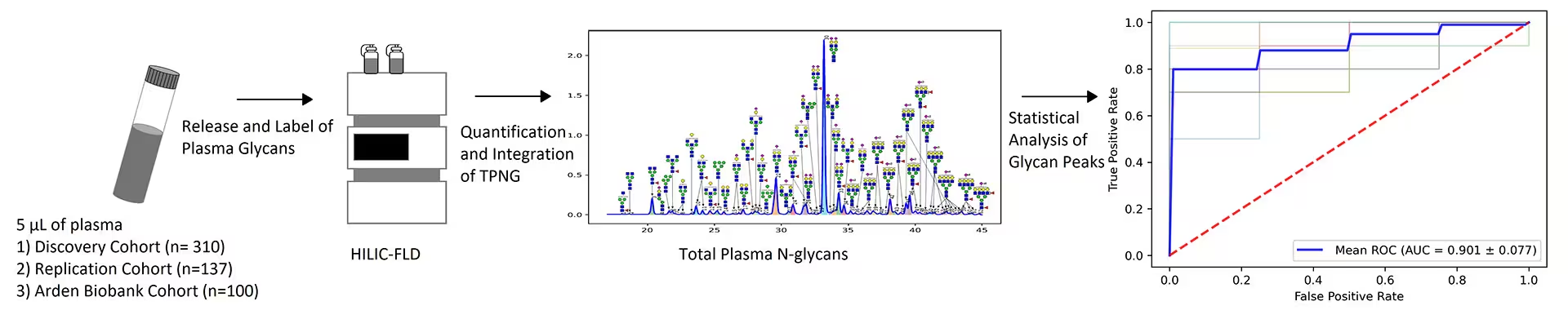
Two glycan peaks showed particularly strong associations: peak 61 (A4G4S4F) was elevated in severe disease, and peak 29 (FA2G2S2) was reduced in severe disease.
These glycomic profiles demonstrated robust prognostic potential, achieving an AUC of 0.826 in the discovery cohort and 0.818 in the independent test cohort.
Together, these findings highlight significant glycoprotein pathway alterations in response to SARS‑CoV‑2 infection and underscore the emerging role of glycan‑based biomarkers in understanding and predicting COVID‑19 outcomes.
Click here to find out more about our exciting biomarker discovery or precision medicine programmes. For more information about this article, visit our Publications webpage.
Ludger a Proud Sponsor of the Extracellular Matrix Biology Across Tissue Sites Meeting

As one of the official sponsors of this year’s “ECM Biology Across Tissue Sites” meeting, Ludger is pleased to support a programme that advances understanding of extracellular matrix biology and fosters collaboration across the research community. This meeting will take place at the University of Manchester from 13th to 14th April 2026.
Click here for more information on this event. Our colleagues, Dr Archana Shubhakar (Head of Business Development) and Carlos Pérez (Scientist) at Ludger, will be attending this event.
If you would like to connect with them at the event for a conversation or learn more about the science we will be presenting, we would be delighted to hear from you. Please get in touch.
Accelerate Confident Alpha Gal Detection with Ludger’s Exoglycosidase Sequencing Workflow
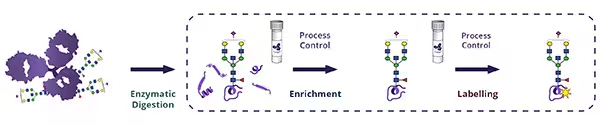
As regulatory expectations tighten around glycosylation control, robust detection of the α Gal (Galα1 3Gal) epitope has never been more critical. Alpha Gal can compromise both the safety and efficacy of biologics, yet its low abundance and structural masking make it notoriously challenging to monitor. Ludger’s proven workflow provides the clarity you need to ensure confident characterisation by combining N glycan release, fluorescent labelling, targeted exoglycosidase digestion, and high-resolution LC-based analysis
Our workflow starts with the release of the glycans from the sample using our PNGaseF Kit, followed by fluorescent labelling with any of our available tags. A clean-up step after labelling removes any excess dye in preparation for the exoglycosidase sequencing.
Next, using enzymes such as α-galactosidase, β-galactosidase, and sialidase enables unambiguous structural confirmation of α-Gal-containing glycans. When combined with HILIC FLR UPLC or LC MS/MS, this workflow delivers sensitive, quantitative insights into even complex mAb glycan populations.
Every step is supported by Ludger’s system suitability standards, reference glycan libraries, and process controls, ensuring reproducibility and regulatory confidence.
Whether you are optimising upstream processes, supporting comparability studies, or preparing regulatory submissions, Ludger’s Alpha Gal workflow empowers you with dependable, high-resolution glycan analysis.
Ready to strengthen your glycoprofiling strategy? Contact us for more information.
Oxford, January 2026
Ludger at Project INTERCEPT’s First Annual Meeting

We are pleased to share that Ludger participated in the 1st Annual Meeting of Project INTERCEPT, which took place on 11–12 December in Amsterdam, hosted by the coordinating institute Amsterdam UMC. This milestone event brought together leading partners from Europe, the United States, and South Korea, marking an important step forward for this ambitious international collaboration.
Over two intensive days, Project INTERCEPT partners came together to dive deep into the science, tackle complex challenges, and co-create innovative solutions aimed at advancing understanding and treatment strategies for Crohn’s disease. The meeting fostered rigorous scientific discussion while encouraging open collaboration, creativity, and long-term strategic thinking as the consortium aligned on its roadmap toward 2026 and beyond.
Ludger was proud to contribute its expertise to this global initiative, engaging alongside academic, clinical, and industry partners united by a shared goal: translating cutting-edge research into meaningful impact for patients. From sparking new ideas and building practical solutions, to strengthening cross-continental partnerships, this first annual meeting laid the foundation for an exciting year ahead.
We look forward to sharing further insights as Project INTERCEPT continues to build momentum and deliver on its promise.
Oxford, December 2025
Glycomics Differentiates Subtypes of Multiple Sclerosis
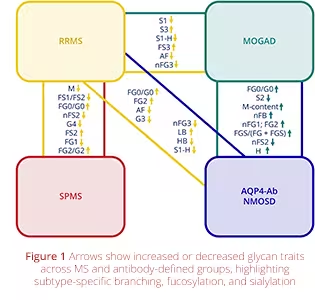
A recent study by T. Kacerova et al., published in Neurology: Neuroimmunology & Neuroinflammation, "Plasma N-Glycan Profiling Enhances Diagnostic Precision in Multiple Sclerosis, AQP4-Ab NMOSD, and MOGAD", showcases the impact of advanced glycomics in understanding multiple sclerosis (MS). The research, led by the Daniel C. Anthony group at the University of Oxford in collaboration with the Daniel I. R. Spencer group at Ludger, used Ludger’s high-resolution mass-spectrometry workflows to map N-glycan profiles in patients with different forms of MS.
The team identified distinct changes in N-glycan branching, sialylation, and fucosylation that separate relapsing-remitting MS from progressive disease, aligning closely with immune activation markers and clinical status. These results demonstrate how glycomics can enable more precise disease stratification and support the discovery of powerful new biomarkers for prognosis and treatment monitoring in neuroinflammatory conditions.
Glycan-Engineered Antibodies Boost Anti-Cancer Immunity

A new study by Alicia M. Chenoweth et al., published in Cancer Research, "An Fc-Engineered Glycomodified Antibody Supports Proinflammatory Activation of Immune Effector Cells and Restricts Progression of Breast Cancer", highlights a powerful role for antibody glycosylation in shaping anti-tumour immunity. The research, led by Professor Sophia Karagiannis and the Cancer Antibody Discovery and Immunotherapy Group at King’s College London, in partnership with Daniel I. R. Spencer’s team at Ludger, used Ludger’s advanced mass-spectrometry glycan profiling to characterise tumour-reactive antibodies in breast cancer.
The team discovered specific N-glycan signatures associated with enhanced immune-cell activation and improved functional responses against tumour cells. These findings deepen our understanding of how antibody glycosylation contributes to cancer immunology, and they underscore the growing potential of glycomics to guide biomarker development and next-generation therapeutic design.
Expanding the Horizons of Glycobiology: Ludger Contributes to Breakthrough Discovery of PNGaseL’s Broad N-Glycan Specificity
A new study directed by Lucy I. Crouch and David N. Bolam, in collaboration with Ludger, has unveiled an exciting breakthrough in glycobiology, demonstrating the unique potential of PNGaseL, an enzyme derived from Flavobacterium akiainvivens. Unlike the widely used PNGaseF, which is limited in the range of N-glycans it can cleave, PNGaseL displays remarkable versatility. It efficiently releases mammalian-, plant-, and invertebrate-type complex N-glycans, as well as high-mannose structures, offering a level of specificity far beyond current tools.
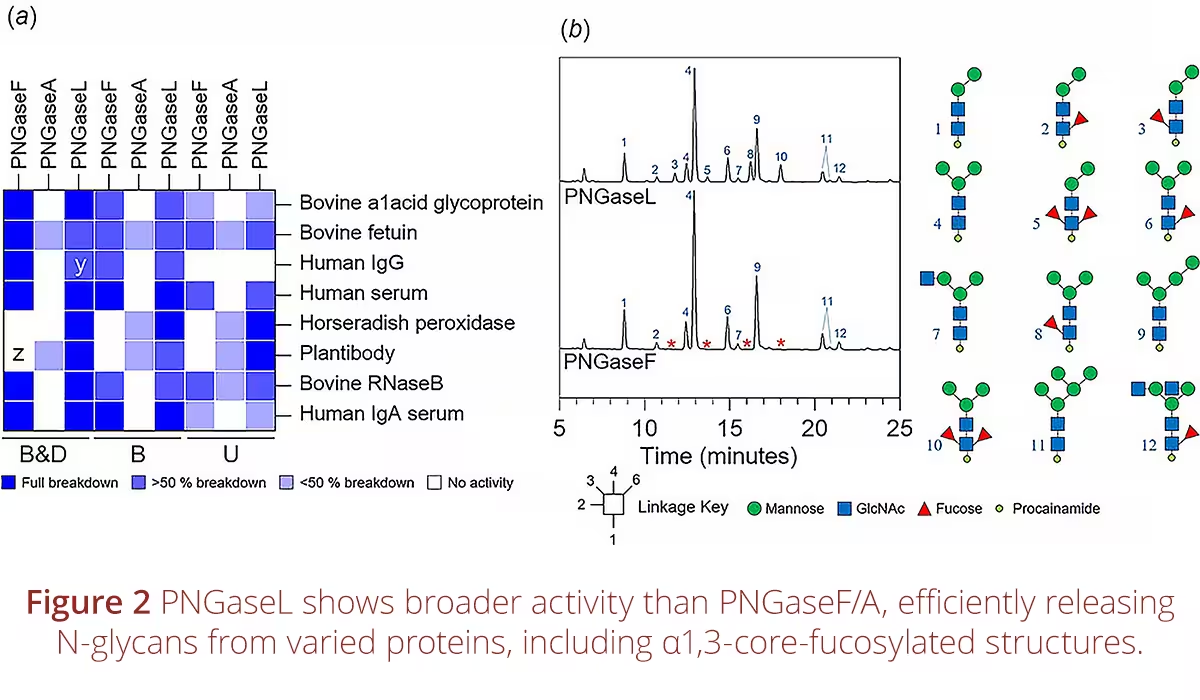
The article, “PNGaseL from Flavobacterium akiainvivens targets a diverse range of N-glycan structures”, provides detailed biochemical and structural analyses, confirming PNGaseL’s stability and broad substrate range. Notably, it functions under standard conditions used for PNGaseF, making it highly compatible with established workflows. The findings position PNGaseL as a next-generation enzyme for advancing glycomics research, with potential to accelerate discoveries in areas ranging from biomarker development to biopharmaceutical quality control.
This collaboration highlights Ludger's leadership in cutting-edge glycobiology, supporting the development of advanced analytical tools that empower researchers worldwide. PNGaseL’s robust performance paves the way for superior glycan profiling, expanding opportunities to explore the N-glycome across species and applications. This innovation marks a significant step forward in understanding glycoproteins and their biological roles.
Industrial Talk Highlights Glycosylation as a Therapeutic Design Tool
The RSC Carbohydrate Group Winter Meeting 2025 took place in York in September, bringing together leading scientists and industry experts to discuss the latest advances in carbohydrate research and its applications.
As part of the industrial sponsorship session, Assoc Prof. Dr. Daniel Spencer delivered an impactful and well received presentation titled "Beyond Decoration – Glycosylation as a Therapeutic Design Tool".
The talk demonstrated the innovative role of glycobiology in enhancing drug performance, with a core focus on the findings of our recent publication, "Enhanced alpha2-3–linked sialylation determines the extended half-life of CHO-rVWF". The presentation illustrated a breakthrough in biotherapeutic design by strategically enhancing the alpha2-3linked sialylation, which successfully extends the therapeutic protein's half-life in the body. This mechanism leverages glycosylation as a powerful design element to significantly improve drug efficacy and performance.
The positive feedback received underscores our commitment to advancing glycoscience and strengthening collaboration between industry and academia!
Oxford, September 2025
Sponsors of RSC Carbohydrate Group Winter Meeting 2025

Ludger Ltd is delighted to be one of the sponsors of the RSC Carbohydrate Group Winter Meeting 2025, taking place 22–23 September in York, UK. This exciting symposium will bring together leading researchers from around the world to share the latest advances in carbohydrate chemistry and foster new collaborations. We look forward to connecting with the community and celebrating innovation in the field.
Personalised Biomolecular Coronas: A New Frontier in Precision Diagnostics
We are proud to announce our latest publication in Analytical Chemistry (Martinez-Serra et al., 2025), showcasing a breakthrough collaboration between our glycomics team and Dr. Marco Monopoli’s nanoscience group at RCSI. Together, we have developed a novel multiple-exposure nanoparticle method that redefines how we study plasma and discover biomarkers.
When blood plasma is repeatedly exposed to fresh silica nanoparticles, layers of “biomolecular coronas” are formed. Early exposures capture abundant clotting and transport proteins such as fibrinogen and apolipoproteins, while later exposures enrich immune and inflammatory proteins, including immunoglobulins and complement factors. This sequential fractionation effectively “peels back” layers of the plasma proteome and glycome, revealing hidden biomarker signatures that are normally masked by highly abundant proteins like albumin.
Using advanced tools such as SDS–PAGE, mass spectrometry, and UHPLC, we traced progressive shifts in both protein and glycan composition across exposures. This creates a personalised corona profile unique to each individual, providing a non-invasive window into health status and disease risk.
By enriching low-abundance biomolecules with diagnostic potential, this method opens new possibilities for precision diagnostics, early disease detection, and patient-tailored healthcare, bringing us closer to a future where monitoring disease is as personalised as the people we treat.
Oxford, July 2025
Ludger at the 16th Jenner Symposium

Ludger was one of the sponsors of the 16th Jenner Glycobiology and Medicine Symposium, held from 11th–13th June 2025 at Maynooth University, Ireland. This global event highlights cutting-edge glycobiology research, with a focus on immunology, infection, cancer, and neurological disorders—areas central to Ludger’s mission of advancing glycoscience.
New Study Reveals Glycosylation Extends CHO rVWF Half-Life

Ludger is proud to be at the forefront of glycobiology, providing advanced tools and expert support that drive pioneering research. A recent publication in Blood titled, “Enhanced α2–3–linked sialylation determines the extended half-life of CHO-rVWF”, was published by Dr. James O’Donnell et al. This research uncovers how specific glycosylation patterns, particularly increased α2,3 sialylation, significantly prolong the half-life of Chinese hamster ovary (CHO) expressed recombinant von Willebrand Factor (rVWF). The research shows that this altered glycosylation reduces β-galactose exposure and clearance via lectin receptors, resulting in enhanced stability and therapeutic potential.
Ludger contributed to this research by providing advanced glycan characterisation through highly sensitive LC-MS analysis, enabling precise identification of the sialylation linkage differences crucial to the study’s conclusions. Although Ludger did not perform lectin-based assays, our analytical expertise and glycan analysis supplied key structural data that supported the validation of glycosylation’s role in improving therapeutic half-life. This collaboration exemplifies how combining academic innovation with industry-leading analytical tools can accelerate the development of more effective biologics.
If you are interested in exploring how Ludger’s technologies and expertise can support your glycosylation research or biotherapeutic development, please contact us at info@ludger.com. We welcome collaborative partnerships that advance scientific discovery and drive innovation in the biopharmaceutical industry.
Ludger was at the 3rd Glycoscience NL Symposium

Ludger was honoured to be a proud sponsor of the 3rd Glycoscience NL Symposium, held on June 27th in Utrecht, Netherlands. This annual event brings together leading experts in glycoscience—including glycobiologists and glyco-chemists—to foster interdisciplinary collaboration and celebrate the diversity of this exciting field.
As part of our sponsorship, Ludger delivered a featured talk, underscoring our ongoing commitment to advancing glycoscience research and its real-world applications.
The day concluded with vibrant networking sessions, strengthening existing partnerships and sparking promising new collaborations.
We remain dedicated to supporting innovation and collaboration in the glycoscience community and look forward to continuing our active engagement in this dynamic scientific arena.
Oxford, May 2025
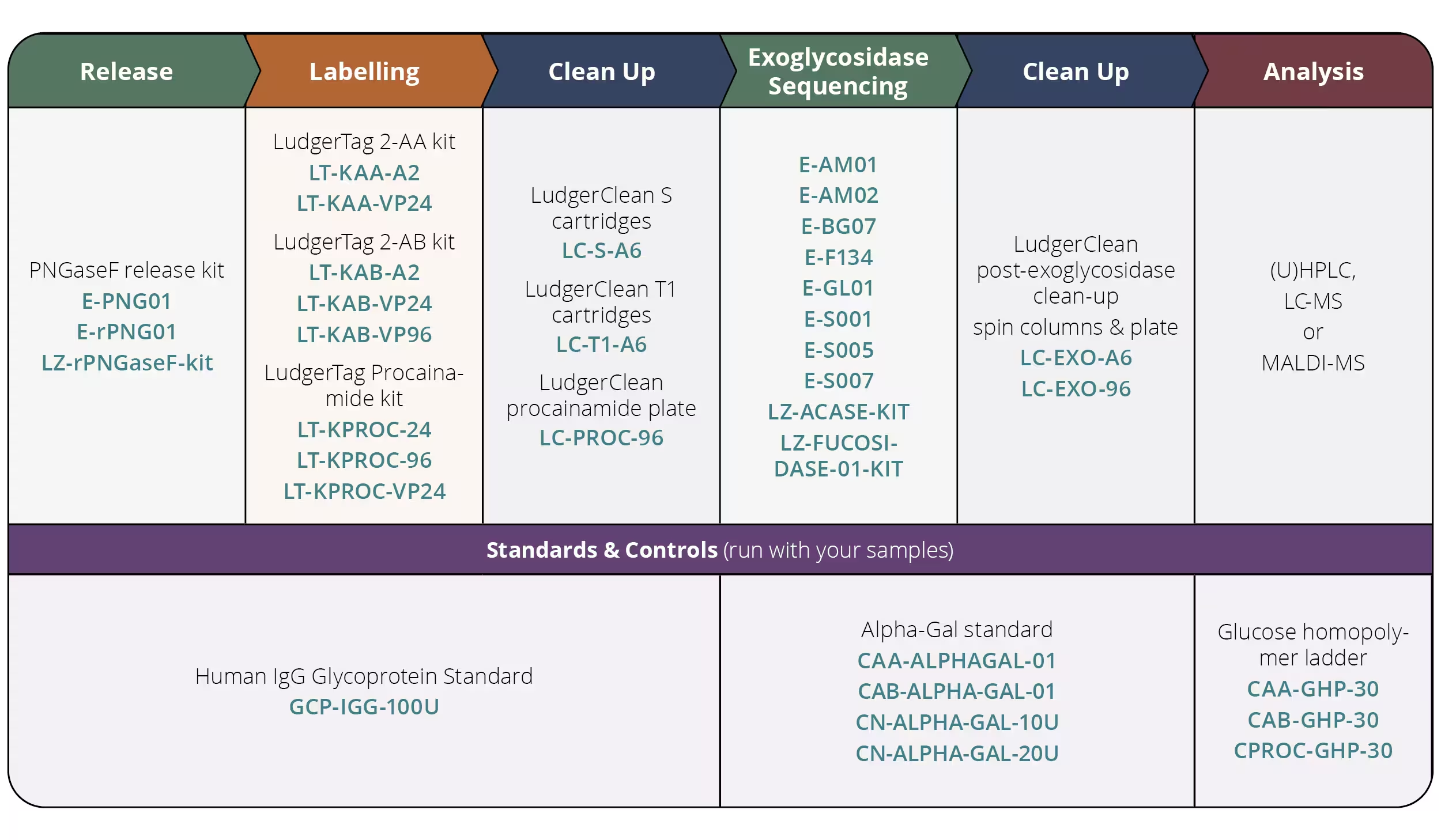
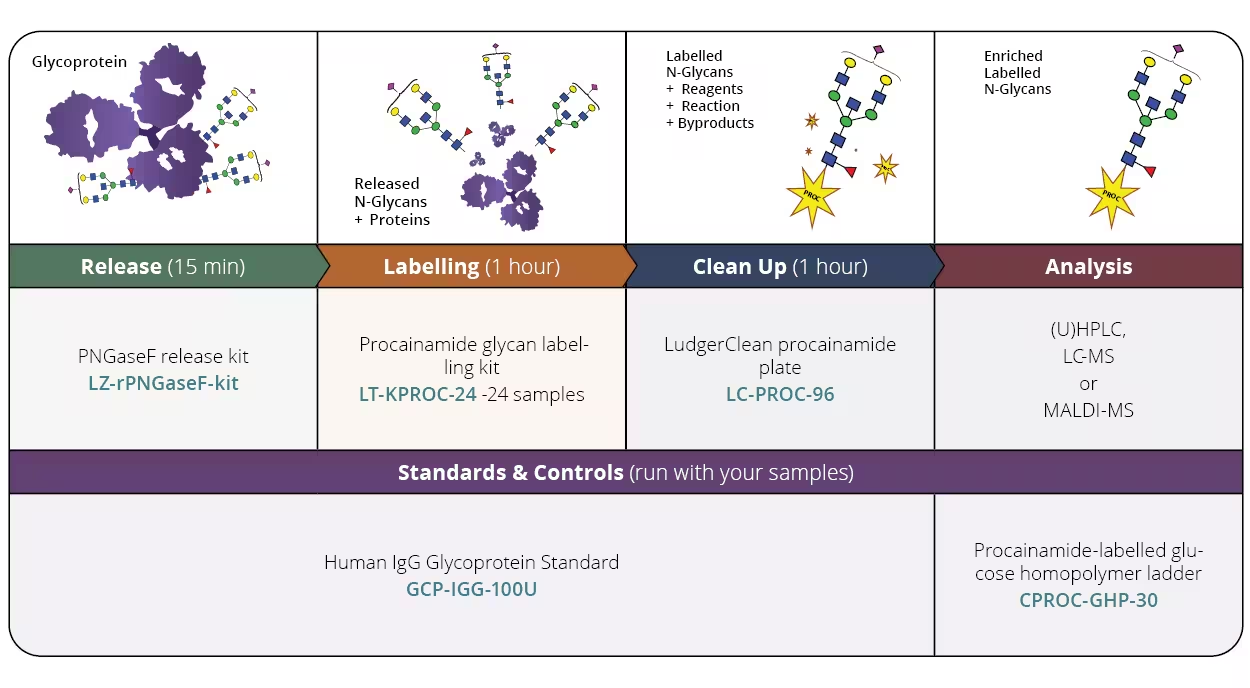
N-Glycan Release & Procainamide Labelling Module Kit
We are excited to launch our Procainamide Release and Labelling Bundle Kit, which includes the complete set of reagents and glycan standards required for N-glycan analysis of therapeutic samples.
LT-KPROC-VP24-MOD is designed to analyse of 24 samples and includes PNGase F enzyme for in-solution release of N-glycans, procainamide labelling kit and a clean-up plate for post-labelling clean-up. The kit also includes an IgG glycoprotein standard to be used as a process control as well as a glucose homopolymer (system suitability standard). The protocol presented is the gold standard methodology for analysing N-linked glycans using (U)HPLC, LC-MS or MALDI-MS approaches.
16th Jenner Glycobiology and Medicine Symposium

Ludger is proud to be one of the sponsors of the 16th Jenner Glycobiology and Medicine Symposium, hosted at Maynooth University, Ireland, from 11th–13th June 2025. This prestigious event brings together leading scientists and clinicians to explore groundbreaking research in glycobiology, with a focus on immunology, infection, cancer, and neurological disorders.
Visit our booth and attend our presentations:
- Poster / Presentation: Potential Sialic Acid and Glycomic Biomarkers of Cardiovascular Disease- Dr Jack Cheeseman et al.
- Poster / Presentation: Nanoparticles as a platform for human plasma prefractionation-Dr Marco P. Monopoli et al.
Meet our experts, discover our latest innovations, and see how Ludger’s cutting-edge glycobiology solutions can support your research. We look forward to connecting with you!