Quantitative Glycan Standards
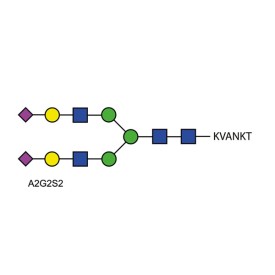
A2G2S2 quantitative glycopeptide standard
BQ-GPEP-A2G2S2-10U
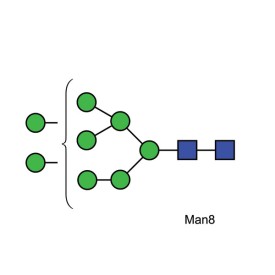
Mannose 8 quantitative standard
BQ-CN-MAN8-10U
Chitotriose quantitative standard (unlabelled)
BQ-CHITOTRIOSE-01
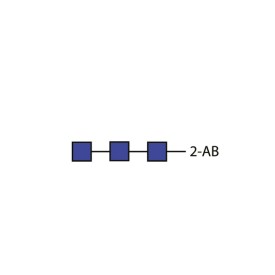
Chitotriose quantitative standard (2-AB labelled)
BQ-CAB-CHI-01
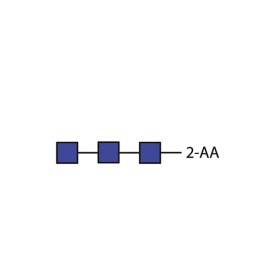
Chitotriose quantitative standard (2-AA labelled)
BQ-CAA-CHI-01
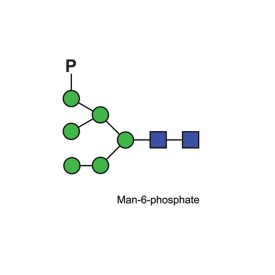
Mannose 6 phosphate quantitative standard
CM-MAN6P-10
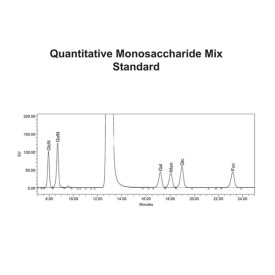
Mix of 6 quantitative monosaccharide standards
CM-MONOMIX-10
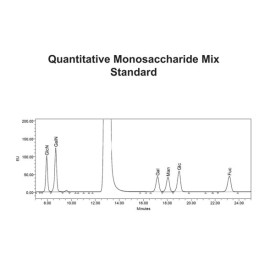
Mix of 6 quantitative monosaccharide standards
CM-MONOMIX-10X3
Xylose quantitative standard
CM-XYLOSE-100
Showing 1 to 9 of 9 (1 Pages)


-275x275.jpg)
-275x275.jpg)

-275x275.jpg)
-275x275.jpg)
-275x275.jpg)
-275x275.jpg)