HILIC Glycan Profiling (Level 1)
Relative glycoprofiling of released N- and O-glycans
The hydrophilic interaction chromatography-(ultra-)high-performance liquid chromatography (HILIC-UHPLC) of released and fluorescently labelled N- and O-glycans is one of the most widely used methods for glycan analysis. The stoichiometric attachment of one label per glycan allows the relative quantitation of different glycan species based on fluorescence and direct comparison of samples to glycan standards leads to preliminary identification of the main glycan species.

This module provides HILIC-UHPLC N- and/or O-glycan profiles with relative quantitation of peaks and preliminary identification of the main glycan species. The analysis will be performed on single or triplicate releases of samples (typically 5-100 µg for each release), an equivalent amount (by volume) of sample buffer negative controls, alongside Ludger positive and negative controls and system suitability standards.
This module is suitable for:
- preliminary glycan assignment
- quality control - profile comparisons to monitor known structures
- monitoring batch to batch consistency
- comparability studies
In order to gain more detailed information on the glycan structures and their relative proportions Level 2 Detailed Characterisation is required.
Sample types:
- Biopharmaceuticals: mAbs, glycoprotein hormones (e.g. follicle stimulating hormone (FSH) and erythropoietin (EPO), Fc fusion proteins, vaccines)
- Cells: mammalian cell lines, bacterial cell components
- Biological fluids, tissues and others
- Glycoproteins set in SDS-gel (N-glycans only)
- COVID-19 patient samples (e.g. plasma, tissues)
- SARS-CoV-2 infected cell lines
Workflow for Level 1 HILIC profiling
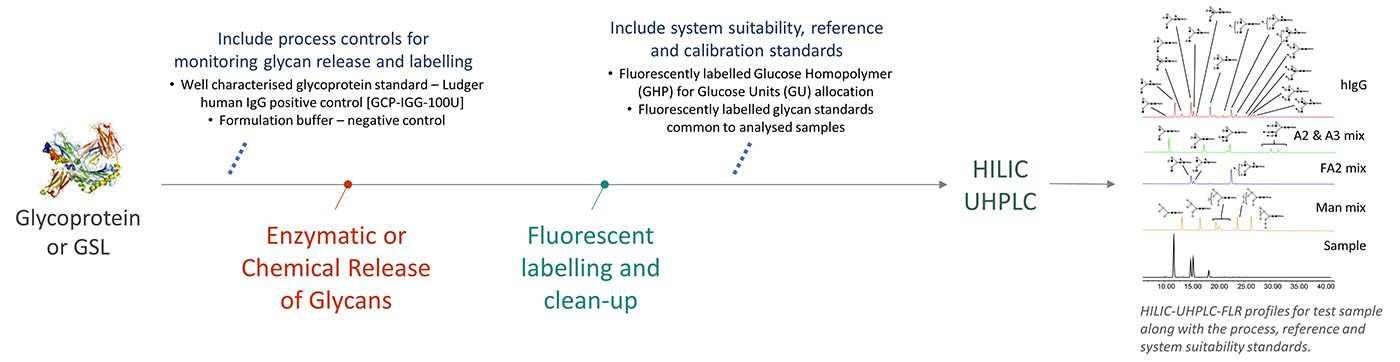
N-glycans are released from glycoprotein by digestion with PNGAse F or PNGAse A.
O-glycans are released from glycoprotein by hydrazinolysis or Orela reagent.
Released glycans are fluorescently labelled with 2-AB or 2-AA or procainamide, purified and analysed by HILIC-HPLC or HILIC-UHPLC.
HILIC-HPLC/UHPLC provides glucose unit (GU) values for profiles where glycan separation is roughly based on glycan size and allows direct profile comparison between samples and standards.
Report
Final report contains:
- HILIC-UHPLC profiles for system suitability standards and Ludger positive controls
- HILIC-UHPLC profiles for client samples and buffer negative controls
- Glucose unit (GU) values
- Relative proportions of detected peaks
- Summary of findings
Comparison of the glycans in client samples with Ludger's glycan standards allows for preliminary identification of the main glycan species
Module Level 1: HILIC profiling has:
4 options for N-glycans profiling to choose between:
- G-L1N-HILIC - Option 1: Glycoprofiling on a single sample releases + formulation buffer + Ludger controls
- G-L1N-HILIC - Option 2: Glycoprofiling on triplicate sample releases + formulation buffer + Ludger controls
- G-X-mAb-LC - Option 1*: High throughput glycoprofiling for up to 43 client samples plus 5 Ludger controls
- G-X-mAb-LC - Option 2*: High throughput glycoprofiling for up to 91 client samples plus 5 Ludger controls
* The sample preparation method will be performed on a Hamilton STARlet liquid handling robot in a 96 well format. This is a low cost solution for analysing a large number of samples in parallel, providing basic chromatographic data on all samples, for comparison purposes.
2 options for O-glycans profiling to choose between:
- G-L1O-HILIC-OHy - Option 1: Glycoprofiling on triplicate sample releases + formulation buffer + Ludger controls.
O-glycan release by hydrazinolysis.
- G-L1N-HILIC-ORe - Option 2: Glycoprofiling on triplicate sample releases + formulation buffer + Ludger controls.
O-glycan release by Orela.

