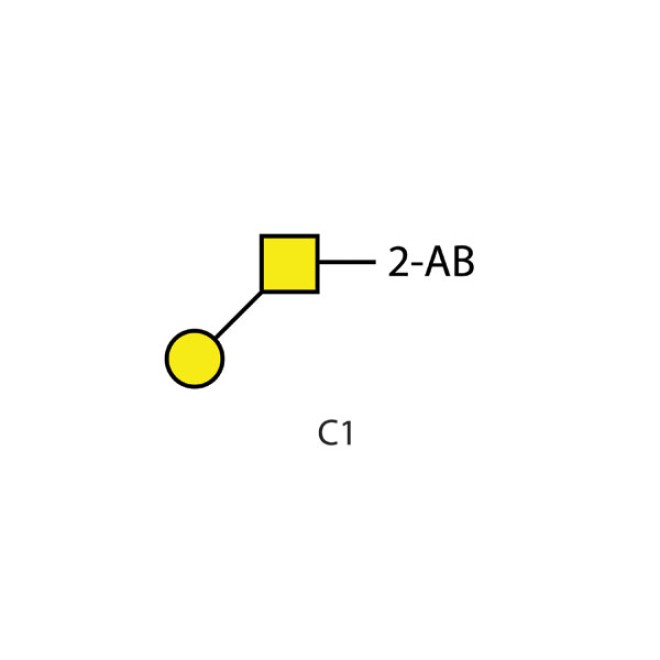
2AB labelled core 1 O- glycan, C1
Core 1 2-AB labelled glycan
Galactose-ß-(1-3)-GalNAc
Linear Core 1 O-glycan with terminal galactose residue, 100pmol.
m/z:503.2115
Product Specification
The 2-AB core 1 glycan consists of a galactose molecule linked β-3 to GalNAc. This glycan is labeled with the fluorescent 2-AB molecule.
Core and sialylated O-glycan standards
O-glycosylation is a common covalent modification of serine and threonine residues of mammalian glycoproteins. The most common type of O-glycosylation found on secreted mammalian glycoproteins and mucins (proteins conjugated to carbohydrate) is from the addition of N-acytylgalactosamine (GalNac) to serine or threonine residues. The Core 1 structure is generated by the addition of galactose b1-3 to this GalNAc. The Core 2 structure is generated by the further addition of N-acetyl-glucosamine b1-6 to the N-acetyl-galactosamine on the Core 1 structure. In total eight different core structures have been identified and all of these can be further elongated by the addition of a number of monosaccharides including sialic acids. These O-glycans are present in biopharmaceuticals such as erythropoeitin (EPO), Etanercept (Enbrel) and human Factor VIII (FVIII). Core 1 and Core 2 O-glycans are also present in biopharmaceuticals such as erythropoeitin (EPO), Etanercept (Enbrel), human Factor VIII (FVIII), Factor IX and insulin glargine. Six 2-AB labelled Core 1 and Core 2 O glycan standards are now available. The standards typically have a purity of > 93% as assessed by HILIC-HPLC and can be used as internal references or system suitability standards for peak assignment during HPLC or UPLC analysis.
Description 2-AB labeled core 1 O-glycan (C1).
Amount 100 picomoles
Form Dry. Dried by centrifugal evaporation from an aqueous solution.
Purity >90% pure as assessed by a combination of CE and HPLC.
Storage Keep at -20°C both before and after dissolution. This product is stable for at least 5 years as supplied.
Shipping The product can be shipped at ambient when dry. After dissolution, ship on dry ice.
Handling Allow the unopened vial to reach ambient temperature and tap unopened on a solid surface to ensure that most of the lyophilized material is at the bottom of the vial. Gently remove the cap, add the desired volume of reconstitution medium, re-cap and mix thoroughly to bring all the oligosaccharide into solution. For maximal recovery of oligosaccharide, ensure that the cap lining is also rinsed and centrifuge the reconstituted vial briefly before use. Ensure that any glass, plasticware or solvents used are free of glycosidases and environmental carbohydrates. Minimize exposure to elevated temperatures or extremes of pH.
Safety This product is non-hazardous and has been purified from synthetic sources certified to be free of all hazardous material including pathogenic biological agents.


-660x660.jpg)
