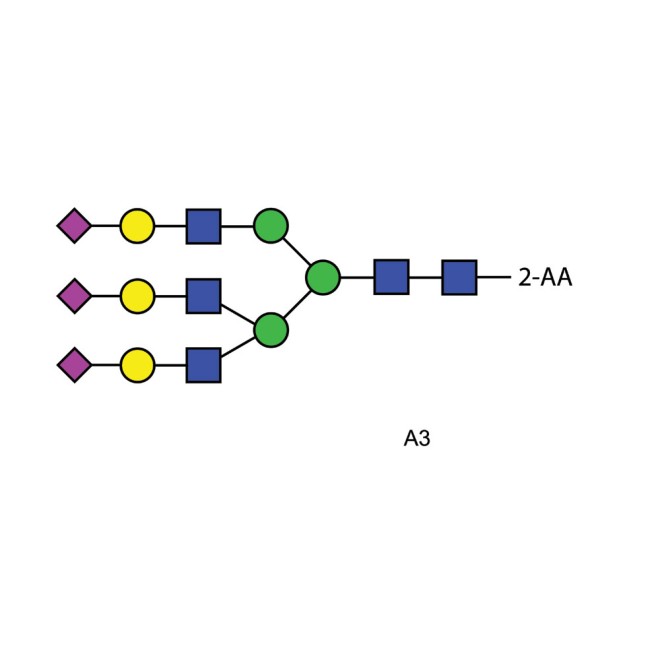
A3 glycan (A3G3S3), 2-AA labelled
2-AA labelled triantennary oligosaccharide. Triantennary N-glycan that contains terminal sialic acid residues, 100pmol.
m/z: 3000.0634
Product Specifications
A3 2-AA Glycan Synonyms A3 N-linked oligosaccharide, A3G3S3
Description mixture of tri-sialylated, tri-antennary N-linked oligosaccharides found on bovine serum fetuin. The two principal components are structural isomers both fully sialylated on the non-reducing terminal galactosyl residues but differing in the distribution of a2,3 and a2,6 linked sialyl residues and the linkage one of the galactoses.
Sources A3 glyans are found on several mammalian glycoproteins including bovine serum fetuin (bSF). This product is typically purified from the oligosaccharide pool released from bSF by hydrazinolysis using a combination of HPLC and glycosidase digestion.
Form Dry. Dried by centrifugal evaporation from an aqueous solution containing approximately 50 mmol dm-3 of ammonium acetate (added to minimize acid-catalysed desialylation).
Molecular Weight 3000
Number of Samples Typically, 100 fmol to 10 pmol of the A3 2-AA glycan can be detected, dependent on the sensitivity of the fluorometer, HPLC or mass spectrometer.
Amount of Sample 100 pmol
Structural Integrity Determined by MALDI mass spectrometry and HPLC.
Labeling Selectivity Essentially stoichiometric labelling. One fluorophore molecule covalently bonded to each glycan.
Detection Fluorescence detection: 360 nm excitation wavelength, 425 nm emission wavelength.
Purity >90% pure as assessed by a combination of 1 H-NMR and HPLC.
Storage -20˚C both before and after dissolution. This product is stable for at least 5 years as supplied. View Certificate of Stabilty
Shipping The product can be shipped at ambient when dry.
Handling Allow the unopened vial to reach ambient temperature and tap unopened on a solid surface to ensure that most of the lyophilized material is at the bottom of the vial. Gently remove the cap, add the desired volume of reconstitution medium, re-cap and mix thoroughly to bring all the oligosaccharide into solution. For maximal recovery of oligosaccharide, ensure that the cap lining is also rinsed and centrifuge the reconstituted vial briefly before use. Ensure that any glass, plasticware or solvents used are free of glycosidases and environmental carbohydrates. Minimise exposure to elevated temperatures or extremes of pH. High temperatures and low pH will cause desialylation. High pH will cause epimerisation of the reducing terminus GlcNAc.
Safety: This product is non-hazardous and has been purified from natural sources certified to be free of all hazardous material including pathogenic biological agents.
HPLC Analysis of the A3 2-AA Glycan
LudgerPure and LudgerTag labeled glycan mixtures may be separated and analysed by a variety of HPLC (high pressure liquid chromatography) methods using LudgerSep™ HPLC columns.
The LudgerSep columns are are available for the following applications:
Separation of charged and neutral glycans via anion exchange columns:
LS-C3-7.5×75 LudgerSep C3 – 7.5x75mm
LS-C2-4.6×50 LudgerSep C2 – 4.6x50mm
Profile analysis of neutral and charged glycans via normal phase columns:
LS-N2-2.0×250 LudgerSep N2 – 2.00x250mm
LS-N2-4.6×250 LudgerSep N2 – 4.6x250mm
LS-N1-4.6×250 LudgerSep N1 – 4.6x250mm
The LudgerSep N2 columns are an especially powerful tool for the purification and analysis of LudgerTag labeled oligosaccharides from complex glycan mixtures. Please contact us for advise regarding your particular application.
Mass Spectrometry and Electrophoresis
LudgerPure and LudgerTag labeled glycans may also be analysed by mass spectrometry, electrophoresis, and various types of spectroscopy. Please call us for advise on the dyes and analysis conditions most suitable for your intended analyses.


-660x660.jpg)
